Description
Uses of Medicine:
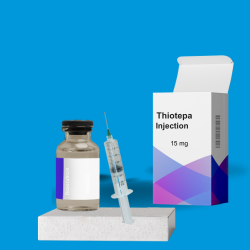
Thiotepa 15 mg/100 mg injection is a chemotherapy drug, an alkylating agent. Patients, Healthcare Professionals (HCP), or Hospitals can buy Thiotepa 15 mg/100 mg Injections online from India through THE INDIAN PHARMA (a trusted Thiotepa Supplier, Exporter, and Contract Manufacturer in India) to:- reduce the risk of graft rejection when used in conjunction with high-dose busulfan and cyclophosphamide as a preparative regimen for allogeneic hematopoietic progenitor (stem) cell transplantation (HSCT) for pediatric patients with class 3 beta-thalassemia.
- treat adenocarcinoma of the breast or ovary.
- control intracavitary effusions secondary to diffuse or localized neoplastic diseases of various serosal cavities.
- treat superficial papillary carcinoma of the urinary bladder.
Like most cancer therapies, Thiotepa injection may cause certain side effects. Some of the most common side effects are diarrhea, skin rashes, blood in the urine, bleeding, and cytomegalovirus infection. Patients may experience these symptoms on behalf of their overall health and dosage regimen. Ensure you notify your doctor about such side effects, particularly when they turn out to be persistent or bothersome to you. Safe and proper administration of medicine, especially during chemotherapy cycles, requires regular health checks and laboratory monitoring.
Before starting treatment with Thiotepa 15 mg/100 mg injection, it is important to let your healthcare provider (HCP) know if you are allergic to it or any of its existing components. It is not advisable to use this medicine if an individual has received live bacteria or virus vaccines like the yellow fever vaccine. Patients with a history of liver, cardiac, renal, and lung diseases should be careful while taking it. People who suffer from bone marrow suppression, which means low red cells (anemia), low white cells (leukopenia), and low platelets (thrombocytopenia), should also take caution.
Regular monitoring of the heart and liver function is required when treating patients with this drug. It is important to note that pregnancy and breastfeeding are contraindications for this medication; accordingly, women of childbearing age must use reliable contraception during the treatment period. Please adhere strictly to all instructions given by your medical advisor when taking Thiotepa Injection.
Dosage & Side Effects:
Class 3 Beta-thalassemia: The recommended dosage of Thiotepa for class 3 beta-thalassemia is two administrations of 5 mg/kg given by intravenous infusion approximately 12 hours apart on Day -6 before allogeneic HSCT, along with high-dose busulfan and cyclophosphamide.Adenocarcinoma of the Breast or Ovary: The recommended dosage of Thiotepa for the treatment of adenocarcinoma of the breast or ovary is 0.3 mg/kg to 0.4 mg/kg by intravenous infusion.
Malignant Effusions: The recommended dosage of Thiotepa for the treatment of malignant effusions is 0.6 mg/kg to 0.8 mg/kg intracavitary.
Superficial Papillary Carcinoma of the Urinary Bladder: The recommended dosage of Thiotepa for the treatment of superficial papillary carcinoma of the urinary bladder is 60 mg in 30 mL to 60 mL of 0.9% Sodium Chloride Injection into the bladder by catheter.
The most common side effects of Thiotepa are neutropenia, diarrhea, mucositis, anemia, thrombocytopenia, hemorrhage, elevated alanine aminotransferase, elevated aspartate aminotransferase, cytomegalovirus infection, hematuria, elevated bilirubin, and rash.