Description
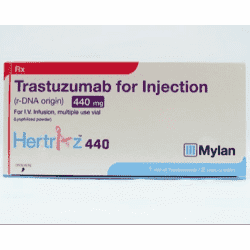
What is Trastuzumab and what it is used for:
Trastuzumab is a HER2/neu receptor antagonist used in the treatment of:
- HER2-overexpressing adjuvant breast cancer.
- First-line treatment of HER2-overexpressing metastatic breast cancer.
- HER2-overexpressing metastatic gastric or gastroesophageal junction adenocarcinoma.
Trastuzumab Dosage:
Trastuzumab 440 mg is available in injection form. Its packaging contains a multi-dose vial of 440mg active ingredient and a vial containing 20mg solvent. The dose of this drug depends upon the stage of cancer and on the General Health of the patient.
Mechanism of Trastuzumab
The HER2 proto-oncogene encodes a transmembrane receptor protein of 185 kDa, which is structurally related to the epidermal growth factor receptor. Herceptin shows the inhibition of the growth of human tumor cells that overexpress HER2. Hertraz is a mediator of antibody-dependent cellular cytotoxicity.
Administration Do not mix Hertraz with other drugs. Do not administer the medicine as an intravenous push or bolus. Treatment of Adjuvant Breast CancerAdminister the drug according to one of the following doses and schedules for a total of 52 weeks of Hertraz therapy:
- An initial dose of Hertraz of 4 mg/kg as an intravenous infusion over 90 minutes, then at 2 mg/kg as an intravenous infusion over 30 minutes weekly during chemotherapy for the first 12 weeks (paclitaxel or docetaxel) or 18 weeks (docetaxel or carboplatin).
- One week succeeding the last weekly dose of Hertraz, administer Hertraz at 6 mg/kg as an intravenous infusion over 30−90 minutes every three weeks.
- As a single agent, administer Trastuzumab within three weeks following completion of multi-modality, anthracycline-based chemotherapy regimens.
- Initial dose at 8 mg/kg as an intravenous infusion over 90 minutes.
- Subsequent doses at 6 mg/kg as an intravenous infusion over 30−90 minutes every three weeks.
Treatment of Metastatic Breast Cancer Administer Hertraz, alone or in combination with paclitaxel, at an initial dose of 4 mg/kg as a 90-minute intravenous infusion followed by subsequent once-weekly doses of 2 mg/kg as 30-minute intravenous infusions until disease progression.
Treatment of Metastatic Gastric Cancer Administer Hertraz at an initial dose of 8 mg/kg as a 90-minute intravenous infusion followed by subsequent doses of 6 mg/kg as an intravenous infusion over 30-90 minutes every three weeks until disease progression.Treatment Reactions:
A common side effect of this drug, when used under some specific diseases are:
- Adjuvant Breast Cancer: headache nausea chills and diarrhea.
- Metastatic Gastric Cancer: diarrhea, anemia, weight loss, neutropenia, stomatitis, upper respiratory tract infection, inflammations, thrombocytopenia, haver, nasopharyngitis, and dysgeusia.
- Metastatic Breast Cancer: Insomnia, Rash, fever, headache, chill, infection, cough, and congestive heart failure.