Description
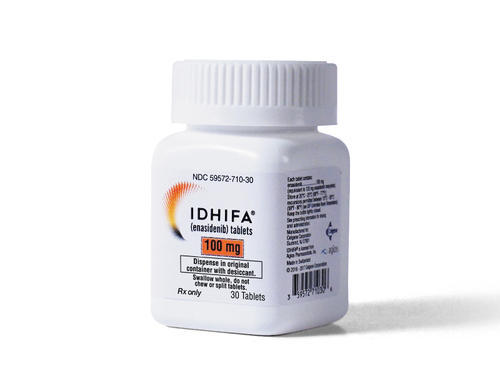
Uses of medication: Idhifa (Enasidenib) is used for the treatment of patients with AML with an IDH2 mutation whose disease has come back or has not improved following previous treatment.
Dosage: The recommended starting dose is idhifa 100mg, which should be administered orally once daily either with or without food about the same time each day. Don’t crush or split the idhifa tablets. If an idhifa dose is puked, missed, or not taken at the usual time, take the dose promptly on the same day, and back to the normal dosing schedule the following day.
Treatment Reactions: The most commonly reported enasidenib side effects include:
- Nausea
- Vomiting
- Diarrhea
- Elevated bilirubin
- Decreased appetite
Warning and precautions
- In patients with idhifa 100mg, if differentiation syndrome is suspected, start corticosteroid therapy and hemodynamic monitoring until the symptom resolution.
- Blood counts, as well as blood chemistries for the leukocytosis and TLS, should be assessed prior to the initiation of enasidenib 50mg.
- Use of idhifa is restricted in females of reproductive potential and males with female partners of reproductive potential.
- Due to potential for adverse reactions in the breastfed infant, women should not breastfeed while on idhifa drug and for at least a couple of months following the last dose.